Abstract
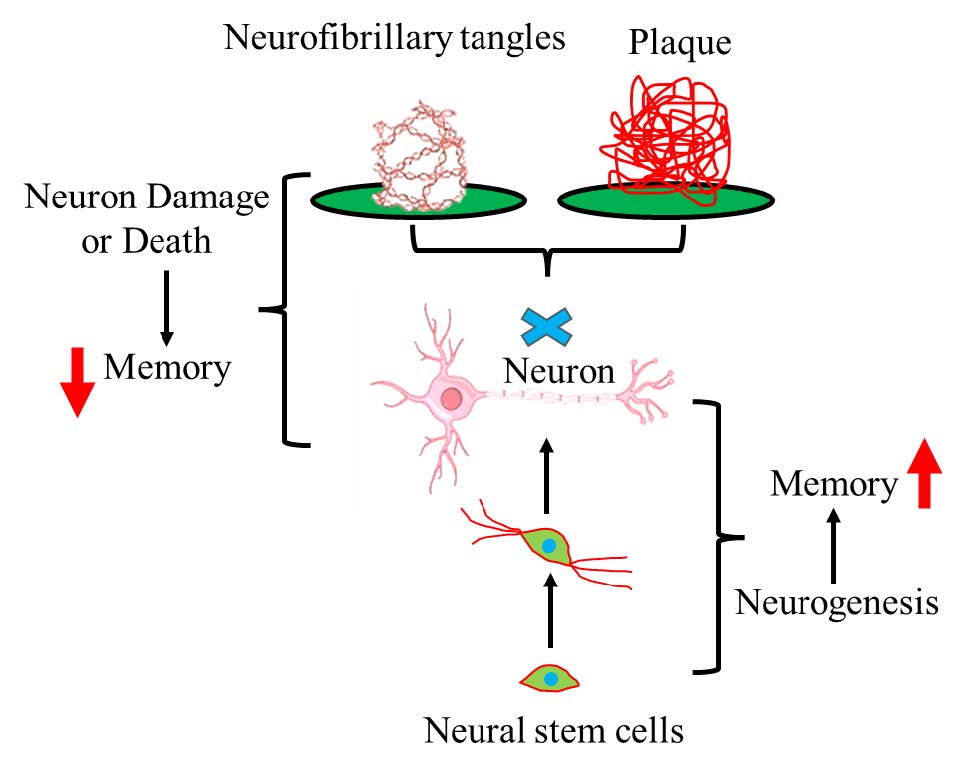
Alzheimer's disease (AD) is increasingly impacting the aging global population, with its prevalence and mortality rates rising steadily. This trend severely affects the quality of life and intensifies the economic strain on both families and society. Despite ongoing research, effective treatments remain elusive. However, enhancing neurogenesis in the hippocampal dentate gyrus (DG) to promote the formation of new neurons and replace those lost in AD patients' hippocampus holds promise as a potential strategy to improve cognitive and memory functions, offering hope for delaying or even treating the disease.
Keywords:Alzheimer's disease; Dentate gyrus; Neurogenesis; Rbm8a gene
Introduction
With the rapid development of the social economy, the phenomenon of population aging has become more and more remarkable, and the prevalence of neurodegenerative diseases closely related to it is also increasing day by day [1-2] . As a neurodegenerative disease, AD is mainly characterized by cognitive impairment and decline in memory ability [3] . With the progression of the disease, patients' language, logical understanding, orientation ability, executive function, and judgment ability will gradually decline [4] . AD is one of the important causes of senile dementia. The incidence rate of dementia patients is 50% - 56%, making it a highly prevalent neurodegenerative disease [5] . According to the World Alzheimer Report 2018, about 50 million people worldwide suffer from dementia [6] . It is estimated that this number will increase to about 82 million in 2030 and about 152 million in 2050 [7-8] . There are more than 200 subtypes of dementia, and dementia caused by AD accounts for 60-80% of all dementias [9] . With the intensification of population aging, the number of AD patients is increasing year by year, and the risk of getting the disease has increased by 14 times. In the United States alone, there are 5.8 million patients, and AD has become the most expensive disease in the United States, with annual medical costs as high as $305 billion [10] . In China, the economic burden of AD is even heavier. It is estimated that by 2050, the economic burden faced by China ($559 billion) will be more than 37 times that in 2011 ($15 billion) [11] . At present, the prevention and treatment of AD have become a global focus. Enhancing neurogenesis to increase the number of new neurons, thereby compensating for the lost neurons in AD, is helpful for improving cognitive and memory abilities and may be an important direction for the future treatment of AD.
Pathological Features of AD
Amyloid Plaque Deposition
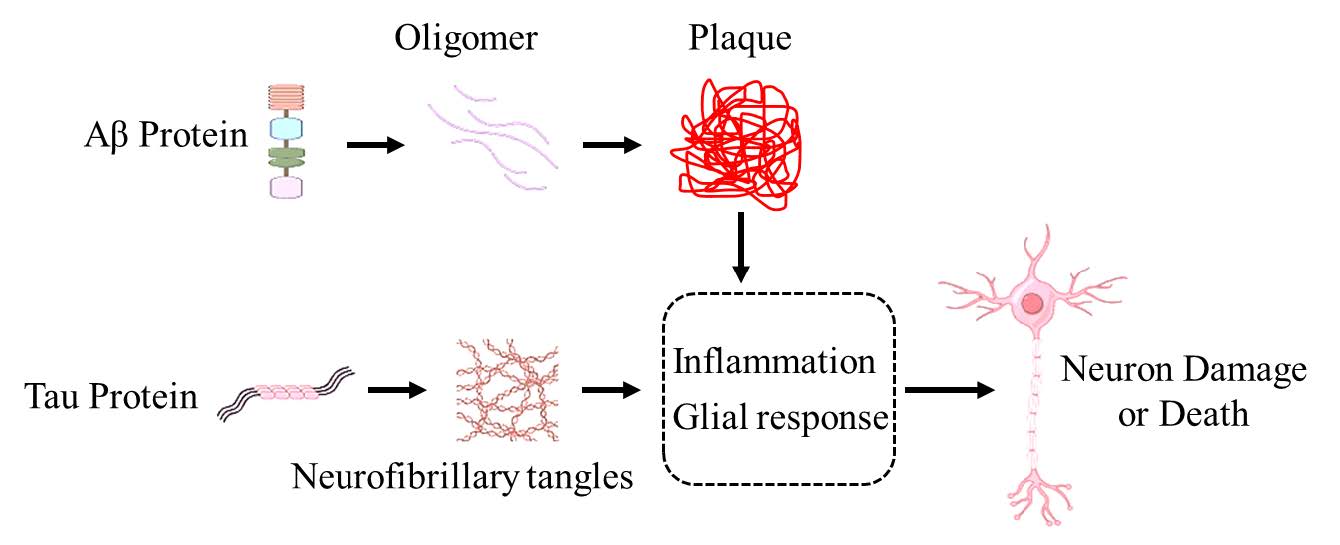
The deposition of amyloid plaques caused by increased production of amyloid β-protein (Aβ) in neurons is one of the main pathological features of AD (Figure 1). Abnormal cleavage of amyloid precursor protein by a series of proteases leads to an imbalance in the secretion of different types of Aβ proteins (mainly Aβ40 and Aβ42), resulting in the production of a large number of neuron-toxic substances and abnormal activation of glial cells, which seriously damage the health of neurons, disrupt their normal structure, and ultimately lead to a large number of neuron deaths and loss of neurites [12-14] .
Figure.1 The formation of Aβ plaque and neurofibrillary tangles. This diagram depicts the pathological features of Alzheimer's Disease (AD). Amyloid β-protein (Aβ) first forms oligomers, which then aggregate into plaques. Tau protein forms neurofibrillary tangles. Both amyloid plaques and neurofibrillary tangles trigger an inflammatory glial response. This response ultimately leads to neuron damage or death, highlighting the key pathological processes underlying AD.
Neurofibrillary Tangles
The pathological features of AD also include the formation of a large number of neurofibrillary tangles by hyperphosphorylated Tau protein in neurons, severe dystrophy of neuronal neurites, and the generation of a large number of neuroinflammatory factors [15] . Among them, amyloid plaque deposition and neurofibrillary tangles first appear in the entorhinal cortex and hippocampus, damaging the neurons in the corresponding parts, even inducing neuron death and synaptic structure destruction, impairing the memory-related neural circuits, and ultimately leading to cognitive impairment and decline in memory ability [16-18] .
Neuron Loss and Treatment Dilemma
The most direct cause of cognitive impairment and memory function decline in AD is the loss of a large number of neurons. Currently, the main methods to protect the normal living environment of neurons include reducing the formation of amyloid plaques, correcting the misfolding of Tau protein, reducing the over - activation of glial cells, and inhibiting the generation of neuroinflammation. However, these methods still cannot effectively delay the progression of the AD disease course and treat AD. Therefore, in addition to reducing the risk factors in the living environment, stimulating the proliferation and differentiation of stem cells to supplement new neurons has gradually become a potentially effective method for treating AD.
Adult Hippocampal Neurogenesis
Definition and Significance of Adult Neurogenesis
Adult neurogenesis mainly refers to the phenomenon that the central nervous system of adult mammals can generate new neurons. The development of new neurons is related to many nervous system diseases. Disrupting adult neurogenesis may lead to various human diseases, including cognitive impairment and neurodegenerative diseases [19-21] .
Process of Adult Neurogenesis
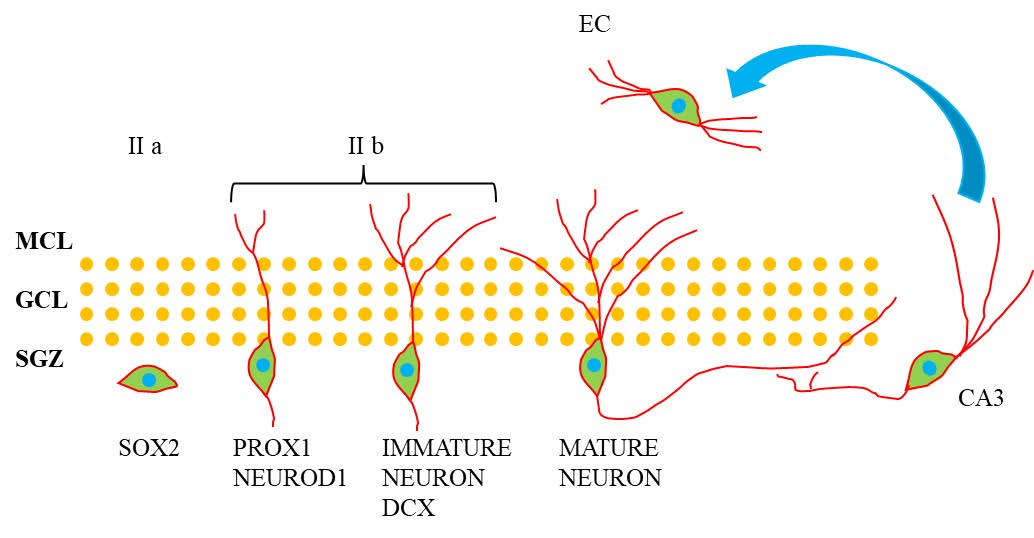
Adult neurogenesis is a multi-stage process in which neural stem cells proliferate and differentiate to generate new neurons and integrate them into neural circuits (Figure 2)[22] . The traditional view is that neurogenesis mainly exists in the embryonic period, gradually decreases with age after birth, and basically disappears in adulthood. However, a large number of studies in the past 30 years have shown that specific brain regions of rodents still have the ability to generate new neurons in adulthood, and this phenomenon is mainly concentrated in the subgranular zone of the hippocampal DG and the subventricular zone adjacent to the lateral ventricle [23-24] . The newly generated neurons in the hippocampal DG play an important role in cognitive function, helping to maintain the integrity of neural circuits and assisting in the storage and transmission of memory. The neural stem cells that promote enhanced neurogenesis are essentially type I radial glial-like cells, which have various states, including quiescent, self-renewing, or proliferative states. Through proliferation and differentiation, they produce intermediate progenitor cells (type II cells) with transient proliferative ability. Type II cells can be further divided into two sub - populations: (1) type IIa cells, which continue to express the neural stem cell marker SOX2; (2) type IIb cells, which are early progenitor cells expressing factors Prox1, NeuroD1, and DCX. Type II cells can produce neural progenitor cells (type III), which then differentiate into mature DG granule neurons. The newly formed neurons in the hippocampal region project their axons to the CA3 region of the hippocampus, and their dendrites enter the outer molecular layer of the DG, forming synapses with the neurons in the second layer of the entorhinal cortex [25-27] . Promoting the generation of new neurons through neurogenesis and integrating them into the hippocampal circuit improves the memory circuit and helps maintain learning and memory abilities. Neurogenesis Defects in AD
Figure.2 The neurogenesis and neural circuit in the dentate gyrus. This diagram illustrates the process of Adult Hippocampal Neurogenesis. Neural stem cells in the subgranular zone (SGZ) of the hippocampus, marked by the SOX2 marker, give rise to progenitor cells. These progenitor cells, indicated by PROX1 and NEUROD1 markers, differentiate into two subtypes (II a and II b). The II b subtype matures into immature neurons, identifiable by the DCX marker. These immature neurons further develop into mature neurons. The granule cell layer (GCL) and molecular cell layer (MCL) are depicted, with the mature neurons eventually integrating into the dentate gyrus circuitry and projecting to the CA3 region. The entorhinal cortex (EC) is shown with an arrow indicating its connection to the hippocampal neurons, highlighting the neural network integration in the process of adult hippocampal neurogenesis.
In 1998, the first evidence of hippocampal neurogenesis in the adult human brain was reported. Throughout life, the DG continuously generates new neurons, endowing the hippocampus with special structural plasticity [28] . However, inhibiting neurogenesis in the hippocampal region leads to a decrease in new neurons and a decline in learning and memory abilities, indicating that neurogenesis in the hippocampal region, which promotes the generation of new neurons, plays an important role in learning and memory [29-30] . Therefore, the cognitive deficits mainly manifested in AD may be related to the decrease in hippocampal DG neurogenesis responsible for learning and memory [31-33] . The specific changes and whether there is damage in the hippocampal DG neurogenesis of AD patients need to be further studied and resolved. Moreno - Jiménez EP et al. compared the immature neurons in the DG of healthy people and AD patients and found that there are thousands of immature neurons in the DG of healthy human subjects, and these neurons show different degrees of maturation during neurogenesis [34] . In contrast, with the progression of AD, the number and degree of maturation of these neurons gradually decrease. This result confirms that neurogenesis persists in the process of human physiological and pathological aging and provides evidence for neurogenesis impairment as a potential mechanism related to memory deficits in AD, which may be a new treatment strategy. To further study the impact of impaired hippocampal neurogenesis on cognitive deficits in AD, Hollands C et al. specifically inhibited hippocampal neurogenesis and found that these animals showed impaired memory behavior after the consumption of neurogenesis [35] . Therefore, the hippocampal region of AD patients does show a certain degree of neurogenesis impairment, which further leads to a decrease in the generation of new neurons and the destruction of the formation of the hippocampal memory circuit, thus explaining the reason for the memory decline in AD patients.
Treating AD by Regulating Neurogenesis
Research has found that the impairment of neurogenesis precedes the pathological changes of AD. Enhancing neurogenesis in the early stage to restore the lost neurons, increase the reserve of normal neurons in the brain tissue, and maintain the integrity of the neural circuit may be a potential treatment strategy for AD [36-37] . Currently, the main ways to enhance neurogenesis include endogenous stimulation of neurogenesis and exogenous stimulation of neurogenesis.
Neurogenesis in the Hippocampal DG and AD
The hippocampal DG is regarded as one of the key brain regions for brain neurogenesis and can be used to explore the purpose of regulating neurogenesis for the treatment of AD. The reasons are as follows: 1. The hippocampus and its neurogenic DG are one of the key brain regions in learning and memory and are also one of the first brain regions to be damaged, mainly manifested as short-term memory loss and disorientation [38-39] . 2. Severe neuron loss in the hippocampal region is another pathological sign of AD. About 1 million neurons are lost in the hippocampal DG of AD patients, and about 5 million neurons are lost in CA1. However, the hippocampal neurogenesis rate in adults is about 700 new neurons per day, and it moderately decreases with age [40] . Therefore, enhancing hippocampal neurogenesis can generate more new neurons, which helps to protect the integrity of the neural circuit, ensure the normal formation and storage of memory, and provides a new treatment direction for neurodegenerative diseases related to hippocampal neuron damage.
Endogenous Stimulation to Promote Neurogenesis
In the Hippocampal DG Throughout adulthood, neural stem cells divide in the stem cell pool, generating progenitor cells and new neurons in the DG of the hippocampus and the subventricular zone. Stem cells maintain the stability of the stem cell pool through self-renewal. However, with age, the ability of neural stem cells to proliferate and differentiate to generate new neurons (i.e., neurogenesis) gradually decreases, leading to neurodegenerative diseases. Enhancing the neurogenesis ability of the hippocampal DG itself through non-stem-cell transplantation is an effective method to correct the decrease in neurogenesis. This method is in line with physiological functions and characteristics and does not cause obvious damage to organ functions and structures.
Exercise Promotes Endogenous Neurogenesis
In recent years, a large number of studies have revealed the important role of exercise in enhancing hippocampal neurogenesis, and its specific mechanism is gradually becoming clear. A research team studied the role of the ErbB4 gene in parvalbumin-positive GABAergic interneurons in mediating the effects of exercise and adult neurogenesis on mental disorders. They found that the inhibitor of the ErbB4 gene significantly impaired a number of memory tasks, including the T-maze, Morris water maze, and contextual fear conditioning, and also reduced the intracellular calcium level in PV neurons. These results are closely related to neurogenesis impairment and can be reversed by exercise, indicating that exercise may participate in regulating adult neurogenesis through the ErbB4 signaling pathway of parvalbumin-positive GABA interneurons, highlighting the importance of exercise in the process of neurogenesis [41] . In addition, another study found similar results. After specifically knocking out the schizophrenia-related gene ErbB4 in PV interneurons, the schizophrenia-related behavioral phenotypes and adult neurogenesis in the hippocampal DG could still be reversed by exercise, further demonstrating that parvalbumin-positive GABAergic neurons play a key role in exercise-mediated adult neurogenesis [42] . To clarify the mechanism throughout the physiological process of neural stem cell proliferation, differentiation, and generation of new neurons in the hippocampal DG, a study reported the proteins co-expressed by progenitor cells, neuroblasts, and mature neonatal neurons in the hippocampal DG and studied their role in neurogenesis. The results showed that the Akt3 protein is involved in the entire neurogenesis process, and the telomerase activity, telomere length, and expression of telomerase reverse transcriptase in Akt3-KO mice were decreased, and the neurite growth of neuroblasts was impaired. The final study found that the Akt3-mTOR signal transduction plays an important role in progenitor cell proliferation and neurite growth [42] . The research team of Frankland PW found that exercise can not only stimulate neurogenesis, promote hippocampal circuit remodeling, and generate new input - output synaptic connections, but also may change the activity pattern of neurons. The change in the activity pattern of neurons over a long-term scale is called representational drift. The research shows that exercise-induced hippocampal circuit remodeling may drive representational drift at the same time [43] . In addition, adiponectin, a cytokine secreted by mature adipocytes, plays an important role in the process of exercise-induced neurogenesis. It can protect the normal proliferation of neural progenitor cells in the hippocampal DG, maintain the dendritic mature morphology of neurons, and ensure the integrity of the neural circuit. This neuro-promoting effect may be achieved through the activation of the AMP-dependent kinase pathway [44] . The research team of Ambrogini P found that in the early life of adult-generated granule cells, three - day physical activity can lead to the early appearance of strong GABAergic synaptic contacts and an increase in the expression of T-type Ca2+ channels. This indicates that three-day voluntary physical exercise can significantly affect the morphological and functional maturation process of new granule neurons when applied in the early stage of their development [45] . However, how to plan physical exercise and sports more carefully to promote neurogenesis scientifically is still worthy of in-depth consideration.
Enriched Living Environment Stimulates Endogenous Neurogenesis In addition to exercise, an enriched environment also helps to promote neurogenesis and enhance the plasticity of the adult hippocampus. Vinogradova A et al. studied the memory behavior and hippocampal neurogenesis of adult male and female Wistar rats. After placing these rats in an enriched environment for two months, they found that both male and female rats that experienced the enriched environment showed an increase in spatial memory ability, and the enriched environment promoted an increase in new neurons in the hippocampal DG of female rats. In male rats, only an increase in the number of proliferating cells and an increase in the expression of brain-derived neurotrophic factors were observed, and no increase in the expression of new neuron markers was detected [46] . Therefore, the enriched environment has a promoting effect on neurogenesis, and the stimulation of new neuron generation by the enriched environment may have gender dependence. Similar studies have found that chronic sleep deprivation can disrupt neurogenesis in the hippocampal DG, and the stimulation of an enriched environment can reverse this process by promoting neuron repair, indirectly indicating the positive regulatory role of an enriched living environment in promoting neurogenesis [47] . However, the specific molecular mechanism by which an enriched living environment improves and stimulates neurogenesis is still unclear and requires a large number of studies to further explore to clarify its scientific nature. Previous studies have suggested that zinc ions are highly concentrated in the distal brain vesicles of mammals, especially in the synaptic vesicles of the hippocampal DG. The research team of Dyck RH studied the role of zinc in neurogenesis and found that zinc in synaptic vesicles is crucial for improving neurogenesis and memory behavior in the adult hippocampus after environmental enrichment, and zinc-containing neurons play an important role in promoting hippocampal plasticity [48] . The enriched environment can not only promote neurogenesis but also change the epigenetic changes in brain function during the aging process, including DNA methylation. The research team of Gerd Kempermann used whole-genome DNA methylation sequencing to find that experiencing a stimulating and enriched environment can offset the age-related DNA methylation changes in the hippocampal DG of mice, confirming the stimulating effect of environmental enrichment on hippocampal plasticity at the DNA methylation level [49] . The study of Hébert JM et al. found that the enriched environment acts autonomously within neurogenic cells through the fibroblast growth factor receptor to expand their number in adult male and female mice, thus acting on neural stem cells and neural progenitor cells and ultimately increasing neurogenesis [50] . Some researchers compared the two different housing environments of an enriched environment and social isolation to explore whether the basic mechanism of cognitive ability changes and synaptic plasticity caused by the housing environment is related to the balance of excitatory/inhibitory synaptic density. The results showed that the living environment may significantly affect the cognitive function, synaptic plasticity, neurogenesis, and neuronal oscillation patterns of mice by changing the balance of excitatory and inhibitory synaptic density, and the enriched living environment has a more positive effect [51] .
Enhancing Endogenous Neurogenesis through Gene Molecular Target Intervention
In addition to exercise and an enriched environment, some drugs targeting specific gene targets have also been found to have a promoting effect on neurogenesis. Previous studies have found that obesity is likely to cause diabetes and is a risk factor for inhibiting neurogenesis. Metformin, a drug for treating pre-diabetes and type 2 diabetes caused by over-nutrition obesity, can restore neurogenesis damage by regulating the composition of the gut microbiota in mice [52] . This process may be achieved by metformin inhibiting the activation of microglia and neuroinflammation. However, it is still unclear which gene targets or targeting molecules this drug uses to participate in the regulation of hippocampal DG neurogenesis. Therefore, more and more studies are beginning to focus on the specific mechanism of neurogenesis, hoping to find molecular targets that regulate neurogenesis, achieve targeted and efficient intervention with specific drugs, and thus enhance hippocampal neurogenesis. More and more studies have shown that positively regulating some gene molecular targets can promote neurogenesis. Beltran-Casanueva R used agonists of neuropeptide Y receptor 1 (NPY1R) and galanin receptor 2 (GALR2) in Sprague-Dawley male rats for intracerebroventricular injection and observed a significant increase in NPY1R-GALR2 co-localization and neuroblast proliferation, as well as enhanced memory consolidation [53] . The author believes that NPY1R and GALR2 are one of the key gene molecular targets for positively regulating neurogenesis and may become potential targets for improving memory-related diseases. Miyata S found through research that the serine protease Motopsin encoded by PRSS12 is also an important target for promoting neurogenesis in the hippocampal DG [54] . Overexpression of this molecule significantly increased the number of proliferating cells and new neurons. In addition, some seemingly harmful substances also have the effect of enhancing neurogenesis. The study found that exogenous administration of the pro-inflammatory cytokine Interleukin-17A (IL-17A) can promote neurogenesis and synaptogenesis in the dentate gyrus (DG), thereby improving behavioral deficits. Moreover, the effect of IL-17A on neurogenesis is more pronounced in females, indicating that the impact of IL-17A is related to gender [55] .
Therefore, apart from appropriate physical exercise and an enriched living environment, effective drug intervention targeting gene molecular targets can also improve hippocampal endogenous neurogenesis. Additionally, enhancing endogenous neurogenesis by identifying gene targets does not pose the ethical issues associated with stem cell transplantation and is more consistent with the original physiological structure. This may be a more promising approach for treating Alzheimer's disease (AD).
Enhanced Neurogenesis by Stem Cell Transplantation
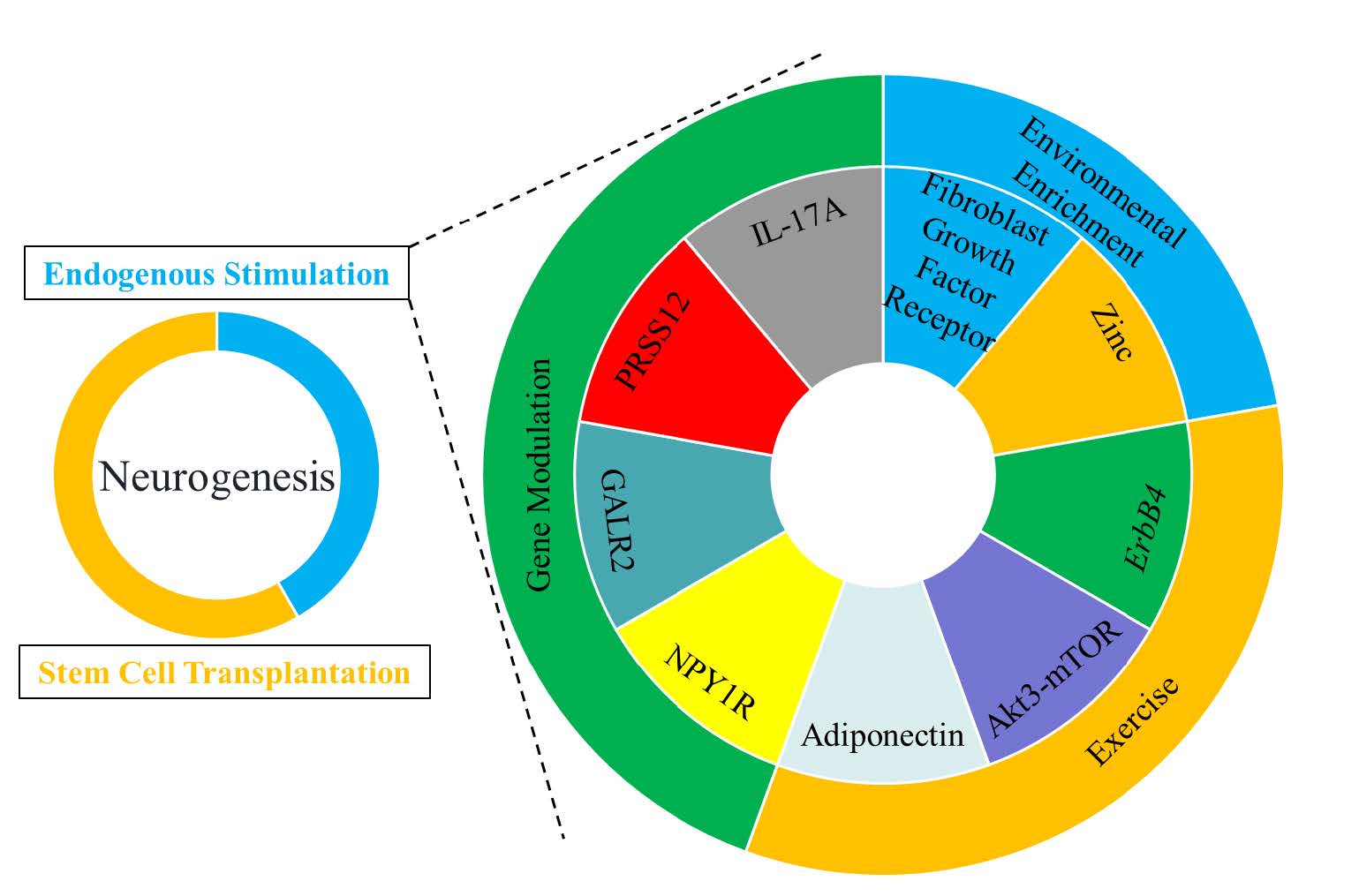
In addition, an increasing number of studies have begun to focus on the exogenous introduction of neural stem cells to restore the neurogenic capacity of AD patients (Figure 3). Generally, several major types of stem cells are used for therapeutic purposes, including embryonic stem cells, neural stem cells, induced pluripotent stem cells, and mesenchymal stem cells. Stem cell therapy has been proposed as a treatment option for neurodegenerative diseases, but the best source of stem cells and their therapeutic effects on neurogenesis remain uncertain. Embryonic stem cells and neural stem cells can effectively generate nerve cells and may be good candidates, but they bring ethical and practical problems. These cells are not only difficult to obtain from good sources, but also often cause immune rejection problems because they may not be autologous cells. Even if the immune rejection problem is overcome, it has been reported that mesenchymal stem cell transplantation can develop into teratomas. Although adult stem cells are more easily accessible, their developmental potential is limited. Authors such as Sugaya K et al. found through research that exosomes can deliver biological materials to adult stem cells to promote the development of mesenchymal stem cells into nerve cells. They also developed a small molecule compound, pyrrolopyrimidine, which significantly increased endogenous neural stem cells through peripheral administration and even eliminated the need for injecting stem cells into the brain [56] . These new technologies may provide neurogenic therapies for AD. An increasing number of studies have begun to recognize that induced pluripotent stem cell technology is helpful for the research of neurodegenerative diseases. Among the challenges in developing drugs for neurodegenerative diseases, the syndromes are usually complex, and small animal disease models cannot fully reproduce the unique characteristics of the human nervous system. Human induced pluripotent stem cells are an ideal new technology, which will allow the generation of nerve cells from individual patients, thus eliminating the species - specific problems inherent in the use of animal models [57-59] . Authors such as Kondo T et al. based on human induced pluripotent stem cell technology found that a cocktail showed significant and powerful anti - Aβ effects on the cells of AD patients. This human induced pluripotent stem cell - based technology platform is expected to be used for the development of AD drugs [60] . Nakano M et al. found that intraventricular injection of bone marrow mesenchymal stem cells could improve the cognitive function impairment of AD model mice by improving astrocyte inflammation and synaptogenesis [61] . Since astrocytes are key cells for synapse formation, the restoration of astrocyte function may lead to synaptogenesis and the correction of cognitive impairment. Similar to the effects of other types of stem cell transplantation, bone marrow mesenchymal stem cell transplantation reduces Aβ deposition and Tau protein phosphorylation, increases neurogenesis, provides support from secreted factors, and improves learning and memory deficits.
Figure.3 Endogenous stimulation and stem cell translation can improve neurogenesis. This circular char tillustrates various factors influencing neurogenesis. The factors are categorized and presented in different colored segments around the circle. Key elements include endogenous stimulation and stem cell transplantation, which are central to the process of neurogenesis. Surrounding these are multiple factors such as genes, environmental factors and exercise. These elements collectively impact neurogenesis, highlighting the complex interplay of biological and environmental components in this process.
In summary, transplanted stem cells can differentiate into new neuron cells and glial cells. By secreting neurotrophic factors and neuroprotective agents, they can stimulate the endogenous repair mechanism and neurogenesis [62-63] . Bone marrow mesenchymal stem cells are relatively easy to obtain, lack immune response and ethical problems, and have good prospects for clinical application [64] . However, the translation from rodent model research to clinical application in the AD patient population is complex. Before the start of clinical trials, key factors such as cell type and source, delivery systems, long-term safety and efficacy, the response of implanted cells to the AD environment, and the mechanism of action need to be systematically and comprehensively evaluated. Therefore, although more and more studies are exploring the role and mechanism of stem cell therapy for AD through animal models, most of the research is still in the exploratory stage.
Therefore, it is particularly important to explore the key genes and targets of impaired neurogenesis in the hippocampal dentate gyrus (DG) of AD. Promoting neurogenesis in the hippocampal DG through endogenous gene regulation is more in line with the physiological structure and function of the organ.
Summary and Prospect
In the past few decades, the field of neurogenesis has thrived. In this review, our understanding of the characteristics of the neurogenic niche, the potential functions of newly formed neurons, and the impact on hippocampal plasticity under physiological and pathological conditions has expanded significantly. However, fundamental challenges remain unresolved. It is still unclear whether specifically improving neurogenesis in AD mouse models can rescue cognitive deficits. Additionally, it is unknown whether neurogenesis in the human brain peaks at a specific point in time or occurs at a constant rate throughout life. Understanding the developmental timeline of neurogenesis and the rate of its activity throughout the life cycle will aid this research in understanding its function in the human brain and in treating diseases. All in all, research on neurogenesis provides new ideas for the treatment of AD, which will help improve the cognitive deficits of AD and delay the progression of the disease.
Acknowledgements
This article is free of any conflict of interest and is written by Chenlu Zhu alone.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1] Kuhn HG, Toda T, Gage FH. Adult Hippocampal Neurogenesis: A Coming-of-Age Story. J Neurosci. 2018;38(49):10401-10410.
[2] Guarnieri FC, de Chevigny A, Falace A, Cardoso C. Disorders of neurogenesis and cortical development. Dialogues Clin Neurosci. 2018;20(4):255-266.
[3] Mercerón-Martínez D, Ibaceta-González C, Salazar C, Almaguer-Melian W, Bergado-Rosado JA, Palacios AG. Alzheimer's Disease, Neural Plasticity, and Functional Recovery. J Alzheimers Dis. 2021;82(s1):S37-S50.
[4] Eratne D, Loi SM, Farrand S, Kelso W, Velakoulis D, Looi JC. Alzheimer's disease: clinical update on epidemiology, pathophysiology and diagnosis. Australas Psychiatry. 2018;26(4):347-357.
[5] Lane CA, Hardy J, Schott JM. Alzheimer's disease. Eur J Neurol. 2018;25(1):59-70.
[6] Raz L, Knoefel J, Bhaskar K. The neuropathology and cerebrovascular mechanisms of dementia. J Cereb Blood Flow Metab. 2016;36(1):172-186.
[7] Hachinski V. Dementia: new vistas and opportunities. Neurol Sci. 2019;40(4):763-767.
[8] Winblad B, Amouyel P, Andrieu S, et al. Defeating Alzheimer's disease and other dementias: a priority for European science and society. Lancet Neurol. 2016;15(5):455-532.
[9] Serrano-Pozo A, Das S, Hyman BT. APOE and Alzheimer's disease: advances in genetics, pathophysiology, and therapeutic approaches [published correction appears in Lancet Neurol. Lancet Neurol. 2021;20(1):68-80.
[10] Alzheimer's Association. 2018 Alzheimer's disease facts and figures[J]. Alzheimer's & Dementia, 2018, 14(3): 367-429.
[11] Keogh-Brown MR, Jensen HT, Arrighi HM, Smith RD. The Impact of Alzheimer's Disease on the Chinese Economy. EBioMedicine. 2015;4:184-190.
[12] Reiss AB, Arain HA, Stecker MM, Siegart NM, Kasselman LJ. Amyloid toxicity in Alzheimer's disease. Rev Neurosci. 2018;29(6):613-627.
[13] Zhang H, Zheng Y. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2019;41(5):702-708.
[14] Reiss AB, Arain HA, Stecker MM, Siegart NM, Kasselman LJ. Amyloid toxicity in Alzheimer's disease. Rev Neurosci. 2018;29(6):613-627.
[15] Quiroz YT, Sperling RA, Norton DJ, et al. Association Between Amyloid and Tau Accumulation in Young Adults With Autosomal Dominant Alzheimer Disease. JAMA Neurol. 2018;75(5):548-556.
[16] Tabatabaei-Jafari H, Shaw ME, Walsh E, Cherbuin N. Cognitive/Functional Measures Predict Alzheimer's Disease, Dependent on Hippocampal Volume. J Gerontol B Psychol Sci Soc Sci. 2020;75(7):1393-1402.
[17] Jaroudi W, Garami J, Garrido S, Hornberger M, Keri S, Moustafa AA. Factors underlying cognitive decline in old age and Alzheimer's disease: the role of the hippocampus. Rev Neurosci. 2017;28(7):705-714.
[18] Balestrieri JVL, Nonato MB, Gheler L, Prandini MN. Structural Volume of Hippocampus and Alzheimer's Disease. Rev Assoc Med Bras . 2020;66(4):512-515.
[19] Sung PS, Lin PY, Liu CH, Su HC, Tsai KJ. Neuroinflammation and Neurogenesis in Alzheimer's Disease and Potential Therapeutic Approaches. Int J Mol Sci. 2020;21(3):701.
[20] Berger T, Lee H, Young AH, Aarsland D, Thuret S. Adult Hippocampal Neurogenesis in Major Depressive Disorder and Alzheimer's Disease. Trends Mol Med. 2020;26(9):803-818.
[21] Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer's disease. Nat Med. 2019;25(4):554-560.
[22] Niklison-Chirou MV, Agostini M, Amelio I, Melino G. Regulation of Adult Neurogenesis in Mammalian Brain. Int J Mol Sci. 2020;21(14):4869.
[23] Disouky A, Lazarov O. Adult hippocampal neurogenesis in Alzheimer's disease. Prog Mol Biol Transl Sci. 2021;177:137-156.
[24] Richetin K, Steullet P, Pachoud M, et al. Tau accumulation in astrocytes of the dentate gyrus induces neuronal dysfunction and memory deficits in Alzheimer's disease. Nat Neurosci. 2020;23(12):1567-1579.
[25] Cope EC, Gould E. Adult Neurogenesis, Glia, and the Extracellular Matrix. Cell Stem Cell. 2019;24(5):690-705.
[26] Kuhn HG, Toda T, Gage FH. Adult Hippocampal Neurogenesis: A Coming-of-Age Story. J Neurosci. 2018;38(49):10401-10410.
[27] Anacker C, Hen R. Adult hippocampal neurogenesis and cognitive flexibility - linking memory and mood. Nat Rev Neurosci. 2017;18(6):335-346.
[28] Anacker C, Hen R. Adult hippocampal neurogenesis and cognitive flexibility - linking memory and mood. Nat Rev Neurosci. 2017;18(6):335-346.
[29] Sahab-Negah S, Hajali V, Moradi HR, Gorji A. The Impact of Estradiol on Neurogenesis and Cognitive Functions in Alzheimer's Disease. Cell Mol Neurobiol. 2020;40(3):283-299.
[30] Dard RF, Dahan L, Rampon C. Targeting hippocampal adult neurogenesis using transcription factors to reduce Alzheimer's disease-associated memory impairments. Hippocampus. 2019;29(7):579-586.
[31] Hollands C, Tobin MK, Hsu M, et al. Depletion of adult neurogenesis exacerbates cognitive deficits in Alzheimer's disease by compromising hippocampal inhibition. Mol Neurodegener. 2017;12(1):64.
[32] Ferreiro E, Lanzillo M, Canhoto D, et al. Chronic hyperglycemia impairs hippocampal neurogenesis and memory in an Alzheimer's disease mouse model. Neurobiol Aging. 2020;92:98-113.
[33] Amber S, Sumera, Mirza FJ, et al. Amyloid-beta Induced Neurotoxicity Impairs Cognition and Adult Hippocampal Neurogenesis in a Mouse Model for Alzheimer's Disease. Curr Alzheimer Res. 2020;17(11):1033-1042.
[34] Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer's disease. Nat Med. 2019;25(4):554-560.
[35] Hollands C, Tobin MK, Hsu M, et al. Depletion of adult neurogenesis exacerbates cognitive deficits in Alzheimer's disease by compromising hippocampal inhibition. Mol Neurodegener. 2017;12(1):64.
[36] Unger MS, Marschallinger J, Kaindl J, et al. Early Changes in Hippocampal Neurogenesis in Transgenic Mouse Models for Alzheimer's Disease. Mol Neurobiol. 2016;53(8):5796-5806.
[37] Choi S H, Bylykbashi E, Chatila Z K, et al. Combined adult neurogenesis and BDNF mimic exercise effects on cognition in an Alzheimer’s mouse model[J]. Science, 2018, 361(6406).
[38] Robinson JL, Molina-Porcel L, Corrada MM, et al. Perforant path synaptic loss correlates with cognitive impairment and Alzheimer's disease in the oldest-old. Brain. 2014;137(9):2578-2587.
[39] West MJ, Coleman PD, Flood DG, Troncoso JC. Differentielt neurontab i hippocampus hos normalt aldrende og hos patienter med Alzheimers sygdom [Differential neuronal loss in the hippocampus in normal aging and in patients with Alzheimer disease]. Ugeskr Laeger. 1995;157(22):3190-3193.
[40] Spalding KL, Bergmann O, Alkass K, et al. Dynamics of hippocampal neurogenesis in adult humans. Cell. 2013;153(6):1219-1227.
[41] Yi Y, Zhang Y, Song Y, Lu Y. Treadmill Running Regulates Adult Neurogenesis, Spatial and Non-spatial Learning, Parvalbumin Neuron Activity by ErbB4 Signaling. Cell Mol Neurobiol. 2024 Jan 29;44(1):17.
[42] Zhang T, Ding H, Wang Y, Yuan Z, Zhang Y, Chen G, Xu Y, Chen L. Akt3-mTOR regulates hippocampal neurogenesis in adult mouse. J Neurochem. 2021 Nov;159(3):498-511.
[43] Yi Y, Song Y, Lu Y. Parvalbumin Interneuron Activation-Dependent Adult Hippocampal Neurogenesis Is Required for Treadmill Running to Reverse Schizophrenia-Like Phenotypes. Front Cell Dev Biol. 2020 Feb 4;8:24.
[44] Wang P, Liang Y, Chen K, Yau SY, Sun X, Cheng KK, Xu A, So KF, Li A. Potential Involvement of Adiponectin Signaling in Regulating Physical Exercise-Elicited Hippocampal Neurogenesis and Dendritic Morphology in Stressed Mice. Front Cell Neurosci. 2020 Jul 3;14:189.
[45] Lattanzi D, Savelli D, Pagliarini M, Cuppini R, Ambrogini P. Short-Term, Voluntary Exercise Affects Morpho-Functional Maturation of Adult-Generated Neurons in Rat Hippocampus. Int J Mol Sci. 2022 Jun 20;23(12):6866.
[46] Vinogradova A, Sysova M, Smirnova P, Sidorova M, Turkin A, Kurilova E, Tuchina O. Enriched Environment Induces Sex-Specific Changes in the Adult Neurogenesis, Cytokine and miRNA Expression in Rat Hippocampus. Biomedicines. 2023 May 2;11(5):1341.
[47] Shixing X, Xueyan H, Yuan R, Wei T, Wei W. Enriched environment can reverse chronic sleep deprivation-induced damage to cellular plasticity in the dentate gyrus of the hippocampus. Transl Neurosci. 2023 Mar 21;14(1):20220280.
[48] Vecchiarelli HA, Patel PP, Hill MN, Dyck RH. Environmental Enrichment Engages Vesicular Zinc Signaling to Enhance Hippocampal Neurogenesis. Cells. 2023 Mar 13;12(6):883.
[49] Zocher S, Overall RW, Lesche M, Dahl A, Kempermann G. Environmental enrichment preserves a young DNA methylation landscape in the aged mouse hippocampus. Nat Commun. 2021 Jun 23;12(1):3892.
[50] rońska-Pęski M, Gonçalves JT, Hébert JM. Enriched Environment Promotes Adult Hippocampal Neurogenesis through FGFRs. J Neurosci. 2021 Mar 31;41(13):2899-2910.
[51] Wang H, Xu X, Xu X, Gao J, Zhang T. Enriched Environment and Social Isolation Affect Cognition Ability via Altering Excitatory and Inhibitory Synaptic Density in Mice Hippocampus. Neurochem Res. 2020 Oct;45(10):2417-2432.
[52] Ma X, Xiao W, Li H, Pang P, Xue F, Wan L, Pei L, Yan H. Metformin restores hippocampal neurogenesis and learning and memory via regulating gut microbiota in the obese mouse model. Brain Behav Immun. 2021 Jul (95):68-83.
[53] Beltran-Casanueva R, Hernández-García A, de Amo García P, Blanco-Reina E, Serrano-Castro P, García-Casares N, Fuxe K, Borroto-Escuela DO, Narváez M. Neuropeptide Y receptor 1 and galanin receptor 2 (NPY1R-GALR2) interactions in the dentate gyrus and their relevance for neurogenesis and cognition. Front Cell Neurosci. 2024 Feb 14; 18:1323986.
[54] Miyata S, Tsuda M, Mitsui S. Overexpression of Motopsin, an Extracellular Serine Protease Related to Intellectual Disability, Promotes Adult Neurogenesis and Neuronal Responsiveness in the Dentate Gyrus. Mol Neurobiol. 2023 Dec 28.
[55] Willinger Y, Friedland Cohen DR, Turgeman G. Exogenous IL-17A Alleviates Social Behavior Deficits and Increases Neurogenesis in a Murine Model of Autism Spectrum Disorders. Int J Mol Sci. 2023 Dec 28;25(1):432.
[56] Sugaya K, Vaidya M. Stem Cell Therapies for Neurodegenerative Diseases. Adv Exp Med Biol. 2018;1056 :61-84.
[57] Penney J, Ralvenius WT, Tsai LH. Modeling Alzheimer's disease with iPSC-derived brain cells. Mol Psychiatry. 2020;25(1):148-167.
[58] Wang C, Ward ME, Chen R, et al. Scalable Production of iPSC-Derived Human Neurons to Identify Tau-Lowering Compounds by High-Content Screening. Stem Cell Reports. 2017;9(4):1221-1233.
[59] mponsah AE, Guo R, Kong D, et al. Patient-derived iPSCs, a reliable in vitro model for the investigation of Alzheimer's disease. Rev Neurosci. 2021;32(4):379-402.
[60] Kondo T, Imamura K, Funayama M, et al. iPSC-Based Compound Screening and In Vitro Trials Identify a Synergistic Anti-amyloid β Combination for Alzheimer's Disease. Cell Rep. 2017;21(8):2304-2312.
[61] Nakano M, Kubota K, Kobayashi E, et al. Bone marrow-derived mesenchymal stem cells improve cognitive impairment in an Alzheimer's disease model by increasing the expression of microRNA-146a in hippocampus. Sci Rep. 2020;10(1):10772.
[62] Tanila H. The role of BDNF in Alzheimer's disease. Neurobiol Dis. 2017;97(1):114-118.
[63] Caffino L, Mottarlini F, Fumagalli F. Born to Protect: Leveraging BDNF Against Cognitive Deficit in Alzheimer's Disease. CNS Drugs. 2020;34(3):281-297.
[64] Nakano M, Kubota K, Kobayashi E, et al. Bone marrow-derived mesenchymal stem cells improve cognitive impairment in an Alzheimer’s disease model by increasing the expression of microRNA-146a in hippocampus[J]. Scientific reports, 2020, 10(1): 1-15.
 Figures
Figures References
References Peer
Peer Information
InformationFigure.1 The formation of Aβ plaque and neurofibrillary tangles. This diagram depicts the pathological features of Alzheimer's Disease (AD). Amyloid β-protein (Aβ) first forms oligomers, which then aggregate into plaques. Tau protein forms neurofibrillary tangles. Both amyloid plaques and neurofibrillary tangles trigger an inflammatory glial response. This response ultimately leads to neuron damage or death, highlighting the key pathological processes underlying AD.

Figure.2 The neurogenesis and neural circuit in the dentate gyrus. This diagram illustrates the process of Adult Hippocampal Neurogenesis. Neural stem cells in the subgranular zone (SGZ) of the hippocampus, marked by the SOX2 marker, give rise to progenitor cells. These progenitor cells, indicated by PROX1 and NEUROD1 markers, differentiate into two subtypes (II a and II b). The II b subtype matures into immature neurons, identifiable by the DCX marker. These immature neurons further develop into mature neurons. The granule cell layer (GCL) and molecular cell layer (MCL) are depicted, with the mature neurons eventually integrating into the dentate gyrus circuitry and projecting to the CA3 region. The entorhinal cortex (EC) is shown with an arrow indicating its connection to the hippocampal neurons, highlighting the neural network integration in the process of adult hippocampal neurogenesis.

Figure.3 Endogenous stimulation and stem cell translation can improve neurogenesis. This circular char tillustrates various factors influencing neurogenesis. The factors are categorized and presented in different colored segments around the circle. Key elements include endogenous stimulation and stem cell transplantation, which are central to the process of neurogenesis. Surrounding these are multiple factors such as genes, environmental factors and exercise. These elements collectively impact neurogenesis, highlighting the complex interplay of biological and environmental components in this process.

[1] Kuhn HG, Toda T, Gage FH. Adult Hippocampal Neurogenesis: A Coming-of-Age Story. J Neurosci. 2018;38(49):10401-10410.
[2] Guarnieri FC, de Chevigny A, Falace A, Cardoso C. Disorders of neurogenesis and cortical development. Dialogues Clin Neurosci. 2018;20(4):255-266.
[3] Mercerón-Martínez D, Ibaceta-González C, Salazar C, Almaguer-Melian W, Bergado-Rosado JA, Palacios AG. Alzheimer's Disease, Neural Plasticity, and Functional Recovery. J Alzheimers Dis. 2021;82(s1):S37-S50.
[4] Eratne D, Loi SM, Farrand S, Kelso W, Velakoulis D, Looi JC. Alzheimer's disease: clinical update on epidemiology, pathophysiology and diagnosis. Australas Psychiatry. 2018;26(4):347-357.
[5] Lane CA, Hardy J, Schott JM. Alzheimer's disease. Eur J Neurol. 2018;25(1):59-70.
[6] Raz L, Knoefel J, Bhaskar K. The neuropathology and cerebrovascular mechanisms of dementia. J Cereb Blood Flow Metab. 2016;36(1):172-186.
[7] Hachinski V. Dementia: new vistas and opportunities. Neurol Sci. 2019;40(4):763-767.
[8] Winblad B, Amouyel P, Andrieu S, et al. Defeating Alzheimer's disease and other dementias: a priority for European science and society. Lancet Neurol. 2016;15(5):455-532.
[9] Serrano-Pozo A, Das S, Hyman BT. APOE and Alzheimer's disease: advances in genetics, pathophysiology, and therapeutic approaches [published correction appears in Lancet Neurol. Lancet Neurol. 2021;20(1):68-80.
[10] Alzheimer's Association. 2018 Alzheimer's disease facts and figures[J]. Alzheimer's & Dementia, 2018, 14(3): 367-429.
[11] Keogh-Brown MR, Jensen HT, Arrighi HM, Smith RD. The Impact of Alzheimer's Disease on the Chinese Economy. EBioMedicine. 2015;4:184-190.
[12] Reiss AB, Arain HA, Stecker MM, Siegart NM, Kasselman LJ. Amyloid toxicity in Alzheimer's disease. Rev Neurosci. 2018;29(6):613-627.
[13] Zhang H, Zheng Y. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2019;41(5):702-708.
[14] Reiss AB, Arain HA, Stecker MM, Siegart NM, Kasselman LJ. Amyloid toxicity in Alzheimer's disease. Rev Neurosci. 2018;29(6):613-627.
[15] Quiroz YT, Sperling RA, Norton DJ, et al. Association Between Amyloid and Tau Accumulation in Young Adults With Autosomal Dominant Alzheimer Disease. JAMA Neurol. 2018;75(5):548-556.
[16] Tabatabaei-Jafari H, Shaw ME, Walsh E, Cherbuin N. Cognitive/Functional Measures Predict Alzheimer's Disease, Dependent on Hippocampal Volume. J Gerontol B Psychol Sci Soc Sci. 2020;75(7):1393-1402.
[17] Jaroudi W, Garami J, Garrido S, Hornberger M, Keri S, Moustafa AA. Factors underlying cognitive decline in old age and Alzheimer's disease: the role of the hippocampus. Rev Neurosci. 2017;28(7):705-714.
[18] Balestrieri JVL, Nonato MB, Gheler L, Prandini MN. Structural Volume of Hippocampus and Alzheimer's Disease. Rev Assoc Med Bras . 2020;66(4):512-515.
[19] Sung PS, Lin PY, Liu CH, Su HC, Tsai KJ. Neuroinflammation and Neurogenesis in Alzheimer's Disease and Potential Therapeutic Approaches. Int J Mol Sci. 2020;21(3):701.
[20] Berger T, Lee H, Young AH, Aarsland D, Thuret S. Adult Hippocampal Neurogenesis in Major Depressive Disorder and Alzheimer's Disease. Trends Mol Med. 2020;26(9):803-818.
[21] Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer's disease. Nat Med. 2019;25(4):554-560.
[22] Niklison-Chirou MV, Agostini M, Amelio I, Melino G. Regulation of Adult Neurogenesis in Mammalian Brain. Int J Mol Sci. 2020;21(14):4869.
[23] Disouky A, Lazarov O. Adult hippocampal neurogenesis in Alzheimer's disease. Prog Mol Biol Transl Sci. 2021;177:137-156.
[24] Richetin K, Steullet P, Pachoud M, et al. Tau accumulation in astrocytes of the dentate gyrus induces neuronal dysfunction and memory deficits in Alzheimer's disease. Nat Neurosci. 2020;23(12):1567-1579.
[25] Cope EC, Gould E. Adult Neurogenesis, Glia, and the Extracellular Matrix. Cell Stem Cell. 2019;24(5):690-705.
[26] Kuhn HG, Toda T, Gage FH. Adult Hippocampal Neurogenesis: A Coming-of-Age Story. J Neurosci. 2018;38(49):10401-10410.
[27] Anacker C, Hen R. Adult hippocampal neurogenesis and cognitive flexibility - linking memory and mood. Nat Rev Neurosci. 2017;18(6):335-346.
[28] Anacker C, Hen R. Adult hippocampal neurogenesis and cognitive flexibility - linking memory and mood. Nat Rev Neurosci. 2017;18(6):335-346.
[29] Sahab-Negah S, Hajali V, Moradi HR, Gorji A. The Impact of Estradiol on Neurogenesis and Cognitive Functions in Alzheimer's Disease. Cell Mol Neurobiol. 2020;40(3):283-299.
[30] Dard RF, Dahan L, Rampon C. Targeting hippocampal adult neurogenesis using transcription factors to reduce Alzheimer's disease-associated memory impairments. Hippocampus. 2019;29(7):579-586.
[31] Hollands C, Tobin MK, Hsu M, et al. Depletion of adult neurogenesis exacerbates cognitive deficits in Alzheimer's disease by compromising hippocampal inhibition. Mol Neurodegener. 2017;12(1):64.
[32] Ferreiro E, Lanzillo M, Canhoto D, et al. Chronic hyperglycemia impairs hippocampal neurogenesis and memory in an Alzheimer's disease mouse model. Neurobiol Aging. 2020;92:98-113.
[33] Amber S, Sumera, Mirza FJ, et al. Amyloid-beta Induced Neurotoxicity Impairs Cognition and Adult Hippocampal Neurogenesis in a Mouse Model for Alzheimer's Disease. Curr Alzheimer Res. 2020;17(11):1033-1042.
[34] Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer's disease. Nat Med. 2019;25(4):554-560.
[35] Hollands C, Tobin MK, Hsu M, et al. Depletion of adult neurogenesis exacerbates cognitive deficits in Alzheimer's disease by compromising hippocampal inhibition. Mol Neurodegener. 2017;12(1):64.
[36] Unger MS, Marschallinger J, Kaindl J, et al. Early Changes in Hippocampal Neurogenesis in Transgenic Mouse Models for Alzheimer's Disease. Mol Neurobiol. 2016;53(8):5796-5806.
[37] Choi S H, Bylykbashi E, Chatila Z K, et al. Combined adult neurogenesis and BDNF mimic exercise effects on cognition in an Alzheimer’s mouse model[J]. Science, 2018, 361(6406).
[38] Robinson JL, Molina-Porcel L, Corrada MM, et al. Perforant path synaptic loss correlates with cognitive impairment and Alzheimer's disease in the oldest-old. Brain. 2014;137(9):2578-2587.
[39] West MJ, Coleman PD, Flood DG, Troncoso JC. Differentielt neurontab i hippocampus hos normalt aldrende og hos patienter med Alzheimers sygdom [Differential neuronal loss in the hippocampus in normal aging and in patients with Alzheimer disease]. Ugeskr Laeger. 1995;157(22):3190-3193.
[40] Spalding KL, Bergmann O, Alkass K, et al. Dynamics of hippocampal neurogenesis in adult humans. Cell. 2013;153(6):1219-1227.
[41] Yi Y, Zhang Y, Song Y, Lu Y. Treadmill Running Regulates Adult Neurogenesis, Spatial and Non-spatial Learning, Parvalbumin Neuron Activity by ErbB4 Signaling. Cell Mol Neurobiol. 2024 Jan 29;44(1):17.
[42] Zhang T, Ding H, Wang Y, Yuan Z, Zhang Y, Chen G, Xu Y, Chen L. Akt3-mTOR regulates hippocampal neurogenesis in adult mouse. J Neurochem. 2021 Nov;159(3):498-511.
[43] Yi Y, Song Y, Lu Y. Parvalbumin Interneuron Activation-Dependent Adult Hippocampal Neurogenesis Is Required for Treadmill Running to Reverse Schizophrenia-Like Phenotypes. Front Cell Dev Biol. 2020 Feb 4;8:24.
[44] Wang P, Liang Y, Chen K, Yau SY, Sun X, Cheng KK, Xu A, So KF, Li A. Potential Involvement of Adiponectin Signaling in Regulating Physical Exercise-Elicited Hippocampal Neurogenesis and Dendritic Morphology in Stressed Mice. Front Cell Neurosci. 2020 Jul 3;14:189.
[45] Lattanzi D, Savelli D, Pagliarini M, Cuppini R, Ambrogini P. Short-Term, Voluntary Exercise Affects Morpho-Functional Maturation of Adult-Generated Neurons in Rat Hippocampus. Int J Mol Sci. 2022 Jun 20;23(12):6866.
[46] Vinogradova A, Sysova M, Smirnova P, Sidorova M, Turkin A, Kurilova E, Tuchina O. Enriched Environment Induces Sex-Specific Changes in the Adult Neurogenesis, Cytokine and miRNA Expression in Rat Hippocampus. Biomedicines. 2023 May 2;11(5):1341.
[47] Shixing X, Xueyan H, Yuan R, Wei T, Wei W. Enriched environment can reverse chronic sleep deprivation-induced damage to cellular plasticity in the dentate gyrus of the hippocampus. Transl Neurosci. 2023 Mar 21;14(1):20220280.
[48] Vecchiarelli HA, Patel PP, Hill MN, Dyck RH. Environmental Enrichment Engages Vesicular Zinc Signaling to Enhance Hippocampal Neurogenesis. Cells. 2023 Mar 13;12(6):883.
[49] Zocher S, Overall RW, Lesche M, Dahl A, Kempermann G. Environmental enrichment preserves a young DNA methylation landscape in the aged mouse hippocampus. Nat Commun. 2021 Jun 23;12(1):3892.
[50] rońska-Pęski M, Gonçalves JT, Hébert JM. Enriched Environment Promotes Adult Hippocampal Neurogenesis through FGFRs. J Neurosci. 2021 Mar 31;41(13):2899-2910.
[51] Wang H, Xu X, Xu X, Gao J, Zhang T. Enriched Environment and Social Isolation Affect Cognition Ability via Altering Excitatory and Inhibitory Synaptic Density in Mice Hippocampus. Neurochem Res. 2020 Oct;45(10):2417-2432.
[52] Ma X, Xiao W, Li H, Pang P, Xue F, Wan L, Pei L, Yan H. Metformin restores hippocampal neurogenesis and learning and memory via regulating gut microbiota in the obese mouse model. Brain Behav Immun. 2021 Jul (95):68-83.
[53] Beltran-Casanueva R, Hernández-García A, de Amo García P, Blanco-Reina E, Serrano-Castro P, García-Casares N, Fuxe K, Borroto-Escuela DO, Narváez M. Neuropeptide Y receptor 1 and galanin receptor 2 (NPY1R-GALR2) interactions in the dentate gyrus and their relevance for neurogenesis and cognition. Front Cell Neurosci. 2024 Feb 14; 18:1323986.
[54] Miyata S, Tsuda M, Mitsui S. Overexpression of Motopsin, an Extracellular Serine Protease Related to Intellectual Disability, Promotes Adult Neurogenesis and Neuronal Responsiveness in the Dentate Gyrus. Mol Neurobiol. 2023 Dec 28.
[55] Willinger Y, Friedland Cohen DR, Turgeman G. Exogenous IL-17A Alleviates Social Behavior Deficits and Increases Neurogenesis in a Murine Model of Autism Spectrum Disorders. Int J Mol Sci. 2023 Dec 28;25(1):432.
[56] Sugaya K, Vaidya M. Stem Cell Therapies for Neurodegenerative Diseases. Adv Exp Med Biol. 2018;1056 :61-84.
[57] Penney J, Ralvenius WT, Tsai LH. Modeling Alzheimer's disease with iPSC-derived brain cells. Mol Psychiatry. 2020;25(1):148-167.
[58] Wang C, Ward ME, Chen R, et al. Scalable Production of iPSC-Derived Human Neurons to Identify Tau-Lowering Compounds by High-Content Screening. Stem Cell Reports. 2017;9(4):1221-1233.
[59] mponsah AE, Guo R, Kong D, et al. Patient-derived iPSCs, a reliable in vitro model for the investigation of Alzheimer's disease. Rev Neurosci. 2021;32(4):379-402.
[60] Kondo T, Imamura K, Funayama M, et al. iPSC-Based Compound Screening and In Vitro Trials Identify a Synergistic Anti-amyloid β Combination for Alzheimer's Disease. Cell Rep. 2017;21(8):2304-2312.
[61] Nakano M, Kubota K, Kobayashi E, et al. Bone marrow-derived mesenchymal stem cells improve cognitive impairment in an Alzheimer's disease model by increasing the expression of microRNA-146a in hippocampus. Sci Rep. 2020;10(1):10772.
[62] Tanila H. The role of BDNF in Alzheimer's disease. Neurobiol Dis. 2017;97(1):114-118.
[63] Caffino L, Mottarlini F, Fumagalli F. Born to Protect: Leveraging BDNF Against Cognitive Deficit in Alzheimer's Disease. CNS Drugs. 2020;34(3):281-297.
[64] Nakano M, Kubota K, Kobayashi E, et al. Bone marrow-derived mesenchymal stem cells improve cognitive impairment in an Alzheimer’s disease model by increasing the expression of microRNA-146a in hippocampus[J]. Scientific reports, 2020, 10(1): 1-15.
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-01-05
Accepted 2025-01-27
Published 2025-02-03


